Class 9 Chemistry important short questions with solved exercise, CRQs, and additional MCQs. Chapter 5 (Energetics) is important for short questions in the board exam.
To read short questions for more chapters, go to the main menu here
The video description is here to understand the full concept.
Q.1 What is the heat and write difference between enthalpy and enthalpy change?
Ans.
| Feature | Heat | Enthalpy |
| Definition | This form of energy is released when a bond is formed and absorbed when it is broken. | It is a measurement of energy in a thermodynamic system. |
| System | It is not an essential part of the system. | It is an essential part of the system. |
| Process | Enthalpy change is equal to the heat evolved or absorbed, at constant pressure. | It is a measurement of energy in thermodynamic system. |
| Measurement | It is a measurement of thermal energy transferred between two objects at different temperatures. | It is a measurement of energy in a thermodynamic system. |
| Representation | It is represented by ‘q’. | It is represented by ‘H’. |
| Unit | Its unit is Joule (J). | Its unit is kilo joules per mol (kJ/mol). |
Enthalpy change: ∆H
The difference between the two enthalpies of a system, indicating the change in terms of heat at constant pressure, is called the enthalpy change.
∆H = H2 – H1
Q.2 Why is breaking a bond an endothermic process?
Ans. Bond breaking is Endothermic. Bond breaking is an endothermic process because when two atoms combine, energy is stored in them, and when we break a bond, an equal amount of energy is given to break that bond. This energy is also known as the dissociation energy. e.g., dissociation energy for hydrogen is 435 kJ/mol.
Q.3 Depict the transition state for the following reaction.
H2+Cl2→2HCl
Ans.
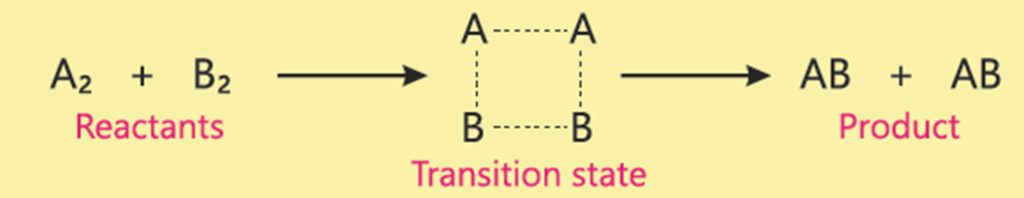
Q.4 Draw the reaction profiles for two exothermic reactions, one of which moves faster than the other. *
Ans.
i. 2H2+O2 → 2H2O
ii. C+O2→CO2
Q.5 What is the role of glycogen in our body?
Ans. Glycogen is the primary storage form of glucose. It is stored in the liver and muscles. It helps to regulate blood sugar levels in our body.
Q.6 Does boiling water in a beaker endothermic or exothermic change? Which form of energy is being transferred in this system? Ans. Boiling water in a beaker is an endothermic reaction. Heat energy is transferred in this system.
Q.7 Can energy be transferred in a form other than heat during a chemical reaction?
Ans. Yes, energy can be transferred in forms other than heat, like light, sound, etc.
Q.8 Why is it not possible to calculate the enthalpy of a system?
Ans. It is not possible to calculate the absolute enthalpy of a system because enthalpy (H) is a state function that depends on the internal energy (E), pressure (P) and volume (V) of a system, we cannot determine the absolute values of these quantities for a system in isolation, instead we calculate the change in enthalpy (ΔH).
Q.9 Why does the chemical reaction between sodium metal and water proceed violently?
Ans. The reaction between sodium and water is exothermic, which is why sodium reacts violently with water. And hydrogen is released during the reaction, which got fire.
Q.10 Is melting of ice an exothermic or endothermic change?
Ans. The melting of ice is an endothermic change.
Q.11 Can an exothermic reaction be reversed?
Ans. Yes, exothermic reactions can be reversed. Hydrogen gas and oxygen gas react to give liquid water in an exothermic reaction.
2H2(g)+O2(g)→2H2O(l) ΔH=−571.6 KJ/mol
571.6 kJ of heat energy is evolved during this reaction. The same amount of energy will be absorbed when the reaction moves in the backward direction, i.e., water will decompose to give hydrogen and oxygen gases back.
2H2O(l)→2H2(g)+O2(g) ΔH=+571.6 kJ/mol
Q.12 Calculate the enthalpy change for the formation of one mole of liquid water.
Ans. Enthalpy change for the formation of one mole of liquid water is:
2H2(g) + O2(g)→ 2H2O(l) ΔH=−571.6KJ/mol
ΔH=-571.6/2 kJ/mol
ΔH=−285.8 kJ/mol
H2(g) + 1/2O2(g)→H2O(l) ΔH=−285.8KJ/mol
The enthalpy of elements in standard states H2 and O2 is zero. The enthalpy of liquid water is -285.8 kJ/mol under standard conditions.
Q.13 Are energy diagrams useful?
Ans. Energy diagrams are useful because it gives us an immediate answer about energy changes happening in a reaction.
Q.14 Draw an energy profile diagram for a hypothetical reaction that does not evolve or absorb heat.
Ans.
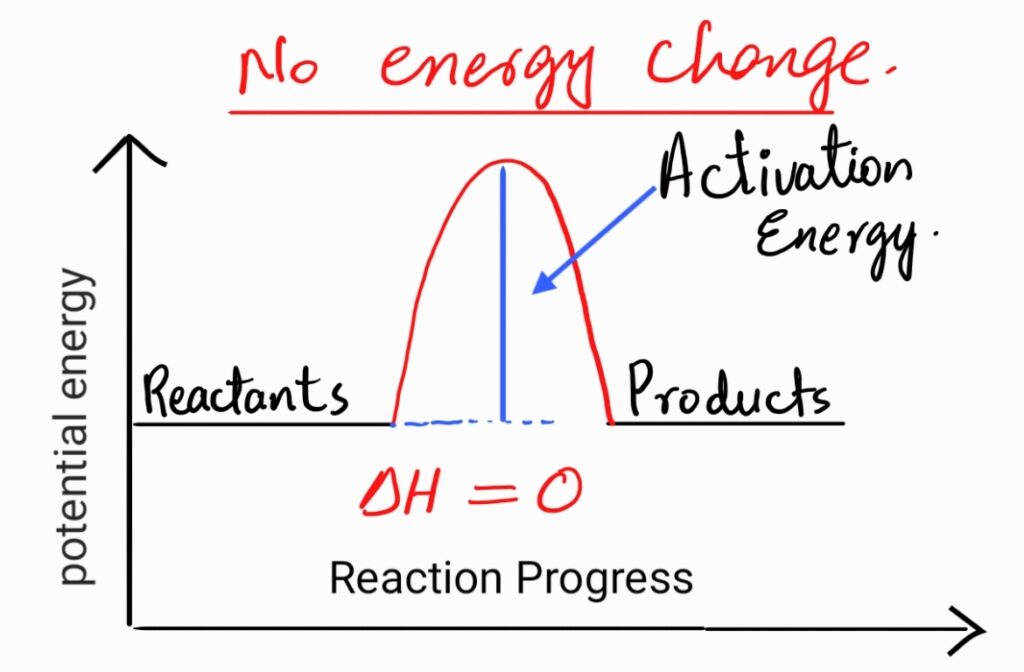
Q.15 Define thermodynamics.
Ans. Thermodynamics deals with how the energy changes during chemical reactions affects the properties of a chemical system.
Q. 16 Who used the word energy for the first time?
Ans. Thomas Young was the first to use the word ‘energy’ in the field of physics in 1802.
Q. 17 What is the difference between Chemical energy and Heat energy?
Ans.
| Chemical Energy | Heat Energy |
| This energy is stored in a molecule in which atoms are bonded to each other. | This form of energy is released when a bond is formed and absorbed when it is broken. |
Q.18 Can we use the energy evolved during a chemical reaction?
Ans. Energy evolved during a chemical reaction is used in everyday for cooking, heating, lighting, transportation, communication, entertainment, and much more.
Q.19 Differentiate between the system and the surroundings.
Ans.
| System | Surrounding |
| In chemistry, any physical or chemical change under study is called a system. | Everything that is not under study and does not fall within the system is called the surrounding. |
| It includes reactants, products, a catalyst, a solvent, and anything else that is important to study for a particular reaction | It includes all other things such as a beaker, burner, test tube, etc. |
Q. 20 What is the importance of enthalpy?
Ans. Enthalpy is important because it tells us how much heat is present in a system. Heat is important because we can extract useful work from it.
Q.21 What is the difference between ΔE and ΔH?
Ans.
| ΔE | ΔH |
| • ΔE is the change in internal energy. | • ΔH is the enthalpy change. |
| • It is equal to the sum of heat and work. | • It is equal to the heat of reaction at constant pressure. |
| ΔE = qv | ΔH = qp |
| • It is equal to the sum of internal energy and the product of pressure volume work. | • It is equal to the sum of internal energy and the product of pressure volume work. |
| • ΔE = q + w | • ΔH = ΔE + P\ΔV |
Q.22 Define standard enthalpy of reaction.
Ans. Standard enthalpy of reaction (ΔHo)
i. “Amount of heat evolved or absorbed when one mole of reactants is converted into products under standard conditions (25∘°C and 1 atm) in a chemical reaction”.
ii. It is represented by (ΔHo).
iii. Its sign is positive for an endothermic reaction and negative for an exothermic reaction.
Reactants and products should be taken in a standard physical state. C(s)+O2(g)→CO2(g) ΔHo=−393.5 kJ (exothermic)
H2(g)+I2(s)→2HI(g) ΔHo=+53.8 kJ (endothermic)
Q. 23 Define the enthalpy of a chemical reaction.
Ans. Enthalpy of Reaction:
The amount of heat or thermal energy evolved or absorbed in a chemical reaction is called the enthalpy of reaction.
Sign: Its sign is negative for exothermic and positive for endothermic reactions.
Q.24 How can atmospheric N2 and O2 react with each other?
Ans. Nitrogen of the atmosphere reacts with oxygen to produce nitrogen oxide (NO) only in the presence of lightning. This is because the reaction is highly endothermic, so only lightning can provide enough energy for this reaction to take place.
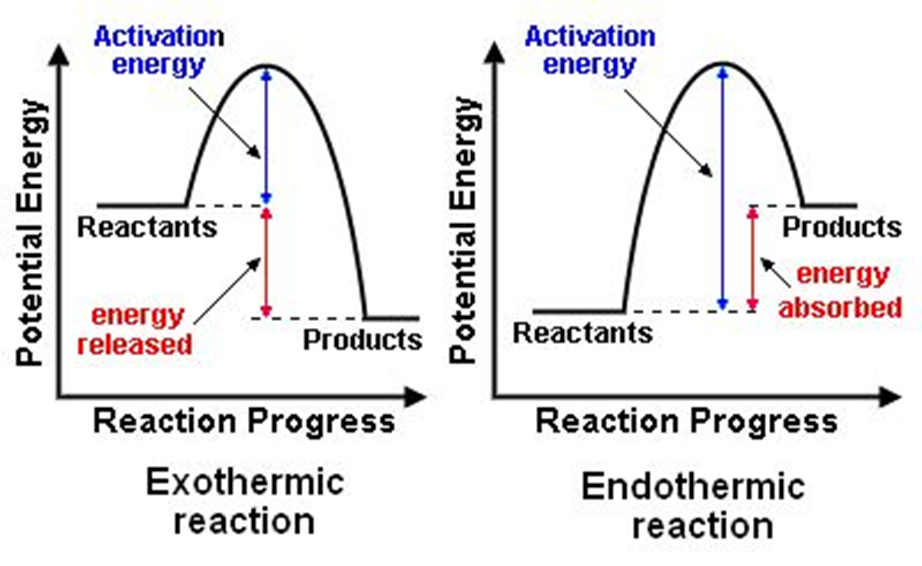
Q.25 Give energy diagrams for endothermic and exothermic reactions.
Ans.

Q.26 Define exothermic and endothermic reactions.
Ans.:
| Exothermic Reactions | Endothermic Reactions |
| A chemical reaction in which heat is released to the surroundings is called an exothermic reaction. | A chemical reaction in which heat is absorbed from the surroundings is called an endothermic reaction. |
| i. C(s) + O2(g) → CO2(g) ΔH° = −393.5 kJ | i. H2(g) + I2(g) → 2HI(g) ΔH° = +53.8 kJ |
| ii. 2H2(g) + O2(g) → 2H2O(l) ΔH° = −571.6kJ | ii. N2(g) + O2(g) → 2NO(g) ΔH° = +180.6 kJ |
Q.27 Give the collision theory of reactions.
Ans. When the two reactant molecules are mixed, all these molecules start colliding with each other. The collisions that result from colliding molecules having average or less than average kinetic energies may not be able to produce any result. But when the two excited molecules from both the reactants collide with each other, they may be able to produce the transition state as shown in the following.
Q.28 Why is the energy of the transition state higher than that of the reactants or products?
Ans.
The energy of the transition state is higher than that of the reactants or products because the bonds between the reactant or product molecules are being cleaved or stretched progressively.
Q.29 How catalyst increase the rate of reaction?
Ans.
The addition of the catalyst in a reaction increases the rate of reaction because it changes the path adopted by the reactants, whereby the activation energy value of the reaction is substantially decreased. As a result, more reactants are now able to be converted into product molecules, and hence the rate of reaction will increase.
Q.30 Define a catalyst with an example.
Ans.
A substance that increases the rate of a chemical reaction without itself undergoing any permanent chemical change.
Example:
i. Nickel (Ni) acts as a catalyst in the hydrogenation of oil to give banaspati ghee.
ii. Platinum (Pt) acts as a catalyst in the production of H2SO4.
iii. Chlorine (Cl2) acts as a catalyst promoting the breakdown of ozone.
Q.31 Draw an energy diagram for the absence and presence of a catalyst.
Ans.
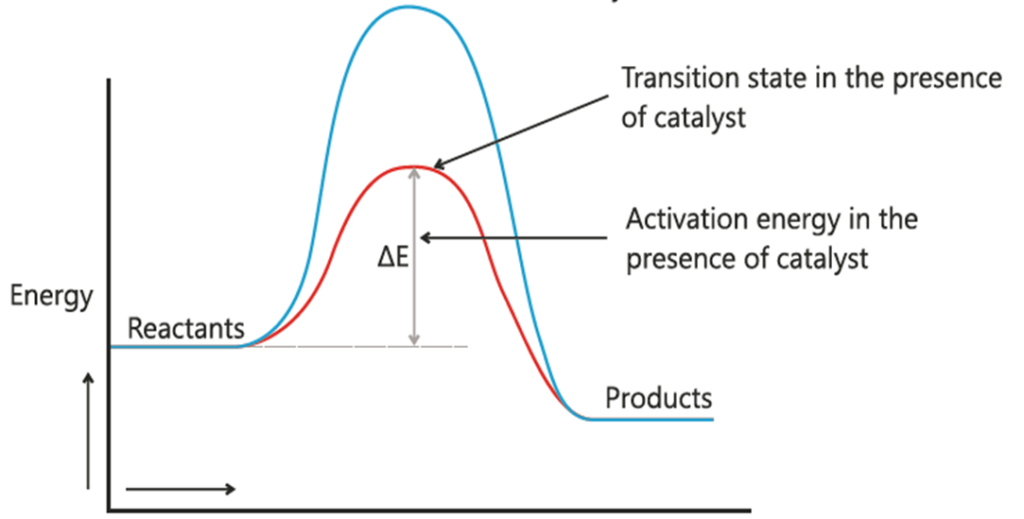
Q.32 Differentiate between aerobic and anaerobic respiration.
Ans.:
| Aerobic Respiration | Anaerobic Respiration |
| ii. Occurs mainly in bacteria, yeast, and some lower organisms. | i. Occurs in the absence of oxygen. |
| ii. Common in higher organisms such as humans and animals. | ii. Occurs mainly in bacteria, yeast and some lower organisms. |
| iii. Complete breakdown of glucose. | iii. Partial breakdown of glucose. |
| iv. End products are CO2 and H2O. | iv. End products are ethanol + CO2 (yeast) or lactic acid (muscles). |
| v. Produces large energy (36–38 ATP). | v. Produces small energy (2 ATP). |
| Example: C6H12O6 + 6O2 → 6CO2 + 6H2O + Energy | Example 1 (Yeast): C6H12O6 → 2C2H5OH + 2CO2 + Energy |
| Example 2 (Muscle cells): C6H12O6 → 2C3H6O3 + Energy |
Q.33 Define Glycolysis.
Ans.
During glycolysis, one molecule of glucose is split into two molecules of pyruvate. This process involves a series of reactions catalyzed by enzymes, with a net production of 2 ATP (Adenosine Triphosphate). When cells of our body require energy for performing metabolic activities, they use this ATP and break it down to get the required energy.
Q.34 How is energy produced from food?
Ans.
The food we eat undergoes digestion in our body, and the digested food molecules that are absorbed by the cells undergo oxidation to produce energy.
Constructed Response Questions
Q.1 (Ex. Q.3 (i)) Physical changes which usually occur around us are given in the table. Write down whether they are exothermic or endothermic. *
Ans.
| Physical change | Exothermic or Endothermic |
| Conversion of hydrated salt into anhydrous salt | Endothermic |
| Burning paper | Exothermic |
| Vaporizing liquid nitrogen | Endothermic |
| Evaporation of dry ice | Endothermic |
| Conduction of electricity by metals | Neither Endothermic Or Exothermic |
| Dissolving ammonium chloride in water | Endothermic |
| Formation of rain from clouds | Exothermic |
| Dissolving sodium carbonate in water | Endothermic |
Q.2 (Ex. Q.3 (ii)) Explain why the reaction between atmospheric gases oxygen and nitrogen does not take place under normal conditions? But in the presence of lightning, these gases react to give NO. The reaction stops as soon as the lightning stops.
Ans. Nitrogen and oxygen do not react under normal conditions because it is a highly endothermic reaction. The nitrogen molecule has a triple covalent bond, which requires very high energy to break. That energy is supplied by lightning. So as long as lightening continue reaction will continue. Equation: N2(g) + O2(g) -> 2NO(g)
Q.3 (Ex. Q.3 (iii)) A reaction between natural gas (CH4) and atmospheric oxygen does not take place when you mix them. As soon as you show a burning match stick, the reaction starts immediately, and then it continues until one or both of the reactions is/are used up. Explain.*
Ans. The activation energy of methane is very high, which is supplied by burning a matchstick. Once the reactants break and combine to form new products, the reaction goes on until one or both reactant get consumed.
Multiple Choice Questions (Exercise)
1. The following reaction is exothermic.
H2 + Cl2 (Sunlight) -> 2HCl.
From where does the energy come to break the bond of H2 and Cl2?
(a) By collisions between the molecules
(b) From sunlight
(c) From the surrounding
(d) By collisions of the molecules with the walls of the container
2. Which of the following reactions has the least value of activation energy?
(a) H2(g) + 1/2 O2(g) -> H2O(g)
(b) C(s) + O2(g) -> CO2(g)
(c) NaCl(aq) + AgNO3(aq) -> AgCl(s) + NaNO3(aq)
(d) H2(g) + I2(s) -> 2HI(g)
3. Which hydrogen halide from the elements is an endothermic reaction?
(a) HCl (b) HF (c) HBr (d) HI
4. What are the products of anaerobic respiration?
(a) ATP + CO2 + H2O
(b) CO2 + H2O
(c) ATP + Ethanol + H2O
(d) Ethanol + CO2 + energy
5. Which reaction do you expect to be a reversible reaction?
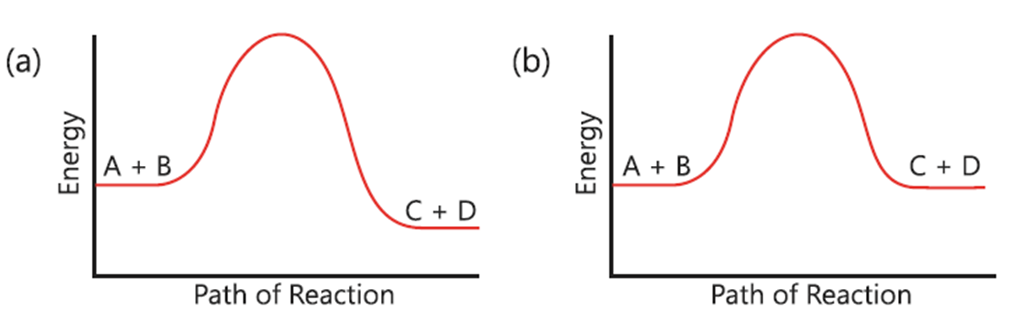
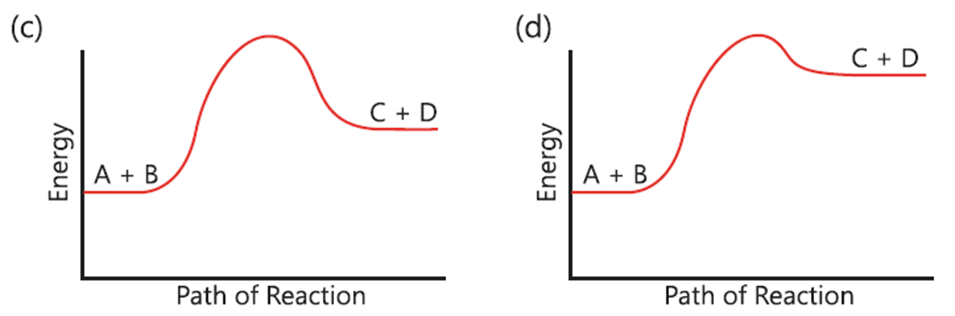
(a) (Reaction profile with a high peak and products lower than reactants)
(b) (Reaction profile showing a lower peak where products can return to reactants)
(c) (Reaction profile with products higher than reactants)
(d) (Reaction profile showing a very high energy barrier)
6. What does it show when a chemical reaction is exothermic?
(a) It shows that the bonds that break are weaker than those that are formed. (b) It shows that the bond which break are stronger than those that are formed.
(c) The exothermic nature of the reaction is not concerned with bond formation or bond breakage.
(d) It shows that the reactants are more stable than the products.
7. When NaOH and HCl are mixed, the temperature increases. The reaction is:
(a) endothermic with a positive enthalpy change.
(b) endothermic with a negative enthalpy change.
(c) exothermic with a positive enthalpy change.
(d) exothermic with a negative enthalpy change.
8. The average bond dissociation energy for the C – H bond is 412 kJ/mol. Which of the following processes will have an enthalpy change close to 412 kJ/mol?
(a) CH4(g) -> C(g) + 2H2(g)
(b) CH4(g) -> C(g) + H2(g)
(c) CH4(g) -> C(g) + 4H2(g)
(d) CH4 -> CH3(g) + H(g)
9. The average bond energies for O-O and O = O are 146 and 496 kJ/mol, respectively. Find the enthalpy in kJ for the following reaction? H-O-O-H(g) -> H-O-H(g) + 1/2 O=O(g)
(a) – 102kJ
(b) + 102kJ
(c) + 3505kJ
(d) + 394kJ
10. Why does the following exothermic reaction not occur?
C (Diamond) -> C (Graphite) ∆H = -3kJ/mol
(a) The structure of diamond is more stable than that of graphite.
(b) Diamond has stronger covalent bonds than graphite.
(c) The change from diamond to graphite has a high activation energy. (d) The density of graphite is less than that of diamond.
—————————————————
SLO-Based Additional MCQ’s
11. When old bonds are broken, the energy is:
(a) Consume (b) Release (c) Remain the same (d) None of these
12. When new bonds are formed, the energy is:
(a) Consume (b) Release (c) Remain the same (d) None of these
13. All chemical reactions involve:
(a) Catalysts (b) Enzymes (c) Energy changes (d) All of these
14. Who used the word energy for the 1st time:
(a) Thomas Young (b) Bohr (c) Rutherford (d) None of these
15. The word energy is used in physics for the first time:
(a) 1802 (b) 1805 (c) 1858 (d) 1902
16. The part of the universe that we want to focus our attention called ____.
(a) system (b) Surrounding (c) Energy (d) Both a & b
17. The enthalpy of reaction C + O2 -> CO2
(a) -571.6 kJ (b) -110.5 kJ (c) -393.5 kJ (d) +53.8 kJ
18. The enthalpy of reaction 2H2 + O2 -> 2H2O
(a) -571.6 kJ (b) -110.5 kJ (c) -393.5 kJ (d) +53.8 kJ
19. The enthalpy of reaction C + 1/2 O2 -> CO
(a) -571.6 kJ (b) -110.5 kJ (c) -393.5 kJ (d) +53.8 kJ
20. The enthalpy of reaction H2 + I2 -> 2HI
(a) -571.6 kJ (b) 11 kJ (c) -393.5 kJ (d) +53.8 kJ
21. If the Delta H value is negative, then the reaction will be:
(a) Exothermic (b) Endothermic (c) May or may not be Exothermic or Endothermic
22. If Delta H is positive, then the reaction will be:
(a) Exothermic (b) Endothermic (c) Both a & b (d) None of these
23. Bond formation energy of one O-H bond is ____:
(a) 484 kJ/mol (b) 486 kJ/mol (c) 488 kJ/mol (d) None
24. Bond dissociation for H2 is:
(a) 430 kJ/mol (b) 435 kJ/mol (c) 440 kJ/mol (d) 445 kJ/mol
25. Bond dissociation for O2 is:
(a) 498 kJ/mol (b) 505 kJ/mol (c) 605 kJ/mol (d) 705 kJ/mol
26. Formation of NO is ____:
(a) Endothermic (b) Exothermic (c) No heat change (d) None of these
27. Activation energy of a chemical reaction must be ____ the average kinetic energy of reacting molecules.
(a) Lower than (b) greater than (c) equal to (d) None of these
28. No reaction occurs if the energy of reacting particles ____ activation energy.
(a) Greater than (b) Lower than (c) Equal to (d) Nearest to
29. Washing clothes at 140F uses almost ____ the energy as at 140F wash:
(a) Half (b) Twice (c) Thrice (d) None of the above
30. ____ of the energy used by traditional electric bulb is wasted in producing heat:
(a) 60% (b) 70% (c) 80% (d) 90%
31. Which is not produced in aerobic respiration?
(a) Carbon dioxide (b) Water (c) Energy (d) Lactic acid
32. ____ acts as reserve energy sources.
(a) Vitamins (b) Proteins (c) Lipids (d) Enzymes
33. Which is released in anaerobic respiration?
(a) Amino acid (b) Stearic acid (c) Citric acid (d) Lactic acid
34. Aerobic respiration releases ____ energy than anaerobic respiration.
(a) Equal (b) less (c) more (d) None of these
35. ____ acts a catalyst promoting the breakdown of ozone.
(a) Cl2 (b) Br2 (c) I2 (d) None
36. During glycolysis, the net ATP produced is:
a) 2 (b) 4 (c) 6 (d) 8




