Class 9 Chemistry important short questions with solved exercise, CRQs, and additional MCQs. Chapter 6 (Equilibria) is important for short questions in the board exam.
To read short questions for more chapters in Class 9 Chemistry, go to the main menu here
Solved Exercise
Multiple choice questions.
- What will happen if the rates of forward and reverse reactions are very high?
- The equilibrium point will reach very soon.
- The equilibrium point will reach very late.
- The reaction will not attain the state of dynamic equilibrium.
- The reaction will be practically irreversible.
- Predict which components of the atmosphere react in the presence of lightening.
- N2 and H2O
- O2 and H2O
- CO2 and O2
- N2 and O2
- An inorganic chemist places one mole of CI2 in a container A, and one mole of each Cl2 and PC13 in container B. Both containers were sealed and heated to the same temperature to reach the state of equilibrium. Guess about the composition of the mixtures in both containers.
- Both containers will have the same composition of mixtures.
- Container A will have more concentration of PCl3 than B.
- Container A will have less concentration of PC13 than B.
- Both the containers will have zero concentration of its reactants.
- CaO or lime is used extensively in steel, glass and paper industries. It is produced in an exothermic reversible reaction by the decomposition of lime (CaCO3). Choose the conditions to produce maximum amount of lime.
- Heating at high temperature in a closed vessel
- Heating at high temperature in an open vessel
- Cooling it in a closed vessel
- Cooling it in an open vessel
- What condition should be met for the reversible reaction to achieve the state of equilibrium?
- All the reactants should be converted into the products.
- 50% of the reactants should be converted into products.
- The concentration of all the reactants and the product should become constant.
- One of the products should be removed from the reaction mixture.
- Why the gas starts coming out when you open a can of fizzy drink?
- Because the solubility of the gas increases
- Because the gas is insoluble in water
- Because the gas is dissolved under pressure hence it comes out when pressure is decreased
- Because the solubility of the gas decreases at high pressure
- The following reaction is performed in an open vessel. How the equilibrium will be affected if you increase the pressure? CaCO3 <===> CaO + CO2
- The forward reaction will be favoured
- The backward reaction will be favoured
- No effect on backward reaction
- No effect on forward or backward reaction
- When a reaction will become a reversible one?
- If the activation energy of the forward reaction is comparable to that of the backward reaction
- if the activation energy of the forward reaction is higher than that of backward reaction
- If the activation energy of the forward reaction is lower than that of backward reaction
- If the enthalpy change of both the reactions is zero.
- Is a reversible reaction useful for preparing compounds on large scale?
- No
- Yes
- They are useful only when equilibrium lies far to the right side
- They are useful only when equilibrium lies far to the left side
- What will happen to the concentrations of the products if a reversible reaction at equilibrium is not disturbed?
- They will remain constant
- They will keep on increasing
- They will keep on decreasing
- They will remain constant for some time and then start decreasing
Questions for Short Answers. (Class 9 Chemistry)
1. How is dynamic equilibrium different from static equilibrium?
Ans: DIFFERENTIATION
The differences between dynamic equilibrium and static equilibrium are as follows:
| Dynamic Equilibrium | Static Equilibrium |
| Definition | |
| “When reaction does not stop only the rates of forward and reverse reaction become equal to each other but take place in opposite directions. This is called dynamic equilibrium state.” Note: Dynamic means reaction is still continuing at dynamic equilibrium state. | “When the system is at rest position, it is called static equilibrium. This happens mostly in physical phenomenon”. |
| Example: | |
| At equilibrium: Rate of forward reaction = Rate of reverse reaction | a building remains standing rather than falling down because all the forces acting on it are balanced. This is an example of static equilibrium. |
Q2. How the following reversible reaction will be affected if its temperature is increased?
2H2O <===> 2H2 + O2
Ans: EFFECT OF TEMPERATURE INCREASE
An increase in temperature will increase the rate of reaction in the above-mentioned example because this is an endothermic reaction
Q3. How can you get the maximum yield in a reversible reaction?
Ans: TO GET MAXIMUM YIELD IN REVERSIBLE REACTION
We can get the maximum yield in a reversible reaction by:
- decreasing pressure if the number of moles of reactants is less than number of moles of products.
- removing products continuously from the reaction mixture
- increasing temperature if the reaction is endothermic
Q4. How can you decrease the time to attain the position of equilibrium in a reversible reaction?
Ans: TO DECREASE TIME TO ATTAIN EQUILIBRIUM
We can decrease the time to attain the position of equilibrium in a reversible reaction by adding a catalyst to the reaction mixture. As the catalyst speeds up both the forward and reverse reactions equally thus equilibrium will be achieved earlier.
Q5. What is the effect of increasing pressure on the following reaction?
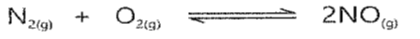
Ans: EFFECT OF INCREASING PRESSURE
There will be no effect of increasing pressure on the given reaction because the number of moles of reactants is equal to the number of moles of products.
CONSTRUCTED RESPONSE QUESTIONS(Class 9 Chemistry)
Q1. Why are some reactions irreversible while others are reversible?
Ans: IRREVERSIBLE AND REVERSIBLE REACTIONS
Some of the reactions are irreversible because the products formed are sufficiently stable under the reaction conditions and are not converted back into the reactants while others are reversible because the products formed are unstable under the reaction conditions and are thus converted back into the reactants again.
Q2. Why are combustion reactions generally irreversible?
Ans: COMBUSTION REACTIONS AS IRREVERSIBLE
Combustion reactions are generally irreversible because once the fuel is burnt due to reaction with oxygen to produce carbon dioxide, water, and heat energy, it becomes quite difficult to use that energy to reverse the reaction and reform the oxygen and original fuel.
Q3. Can you make an irreversible reaction reversible and vice versa?
Ans: i. Making an irreversible reaction reversible.
Some irreversible reactions can become reversible by changing reaction conditions (temperature, pressure, or catalyst). For example. The decomposition of calcium carbonate can become reversible if carbon dioxide is kept at high pressure.
ii. Making a reversible reaction irreversible.
A reversible reaction can be made irreversible by removing products continuously. (e.g., releasing gas, forming precipitate). For example, removing ammonia gas from the Haber process forces the reaction forward.
Q4. How do you know if a reaction is reversible or irreversible?
Ans: REVERSIBLE OR IRREVERSIBLE REACTION
Irreversible Reaction
A reaction is an irreversible chemical reaction if it can occur in only one direction. The reactants can change to the products, but the products cannot change back to the reactants.
Reversible Reaction
Reversible chemical reactions can occur in both directions. The reactants can change to the products, and the products can also change back to the reactants
Q5. Are the phase changes in water (solid to liquid, liquid to vapour) reversible or irreversible?
Ans: PHASE CHANGE IN WATER
Phase changes in water (solid to liquid, liquid to vapor) are reversible. It is purely a physical phenomenon. For example
- Ice (solid) melts to liquid water, and water can freeze back to ice.
- Water (liquid) evaporates to vapour, and vapour condenses to water again.




